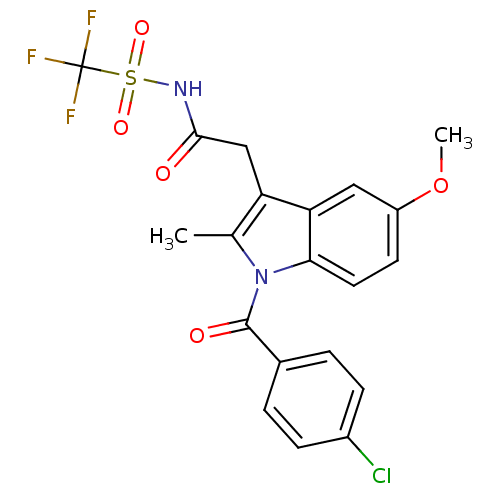
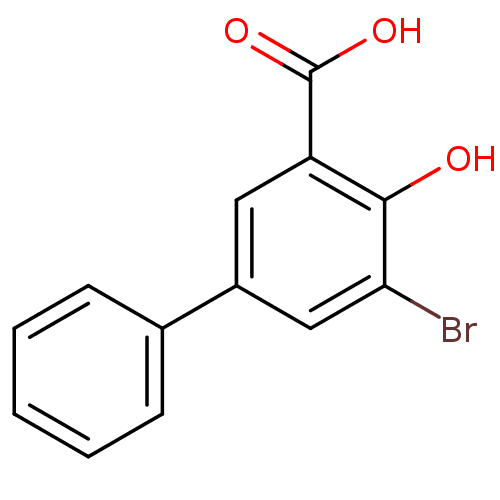
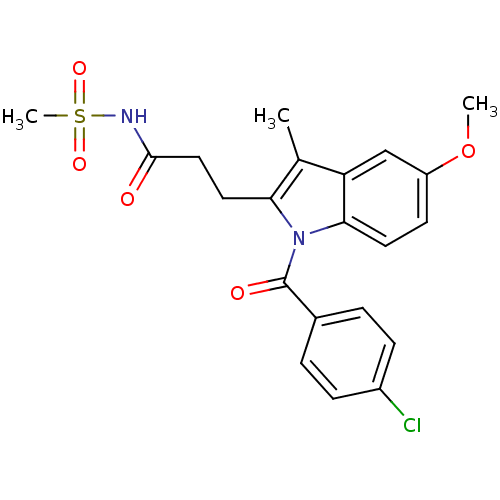
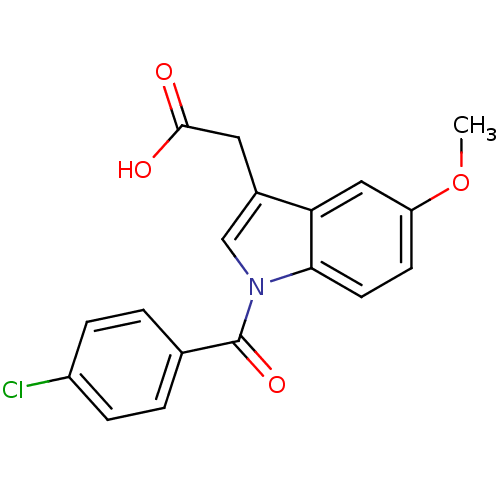
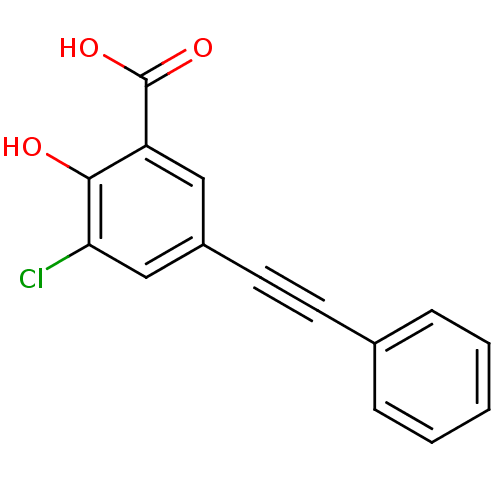
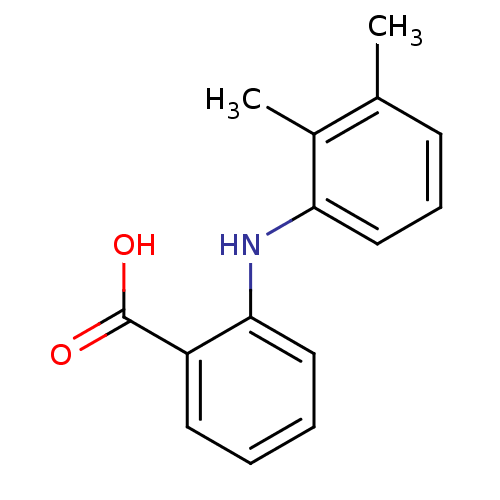
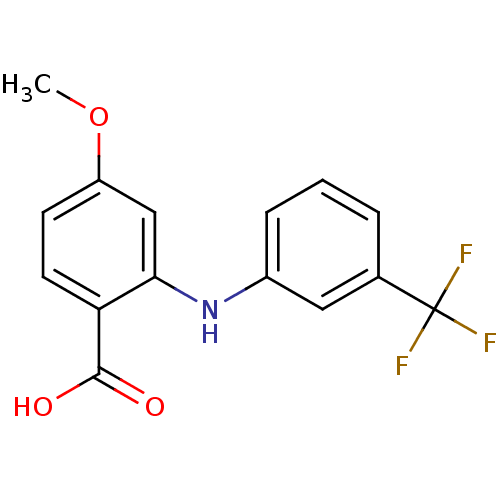
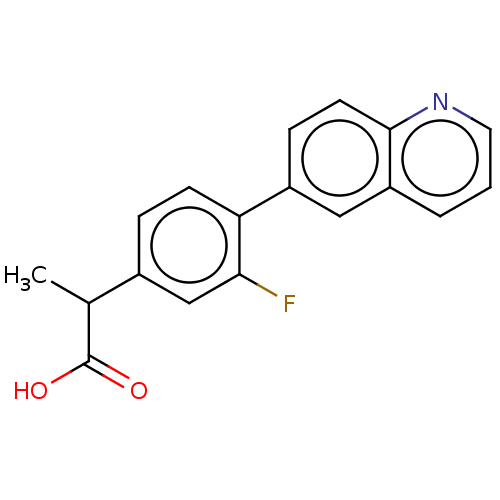
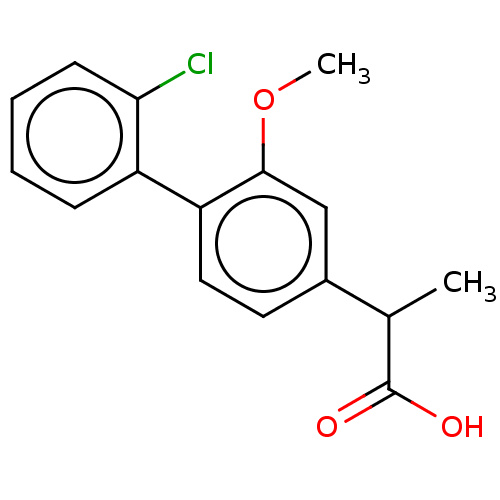
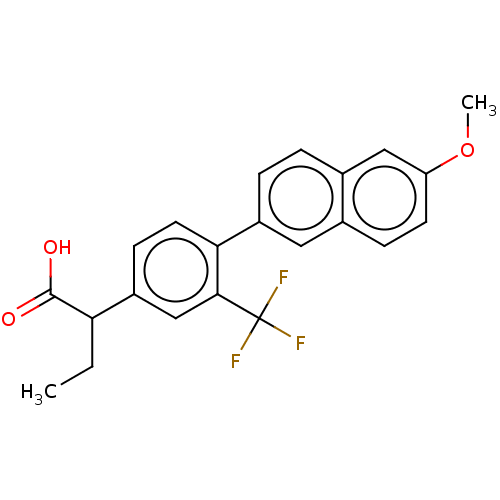
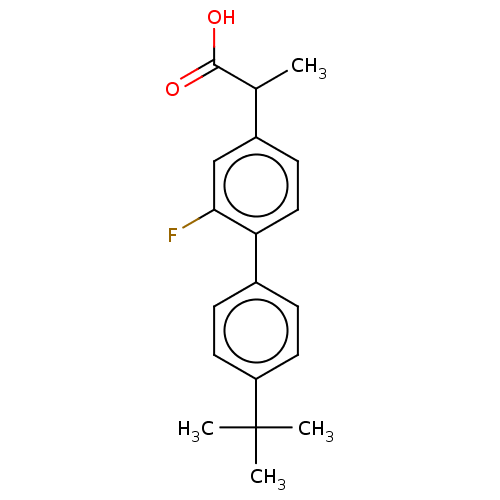
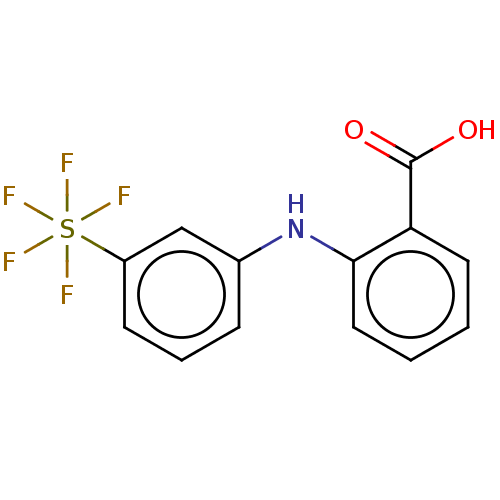
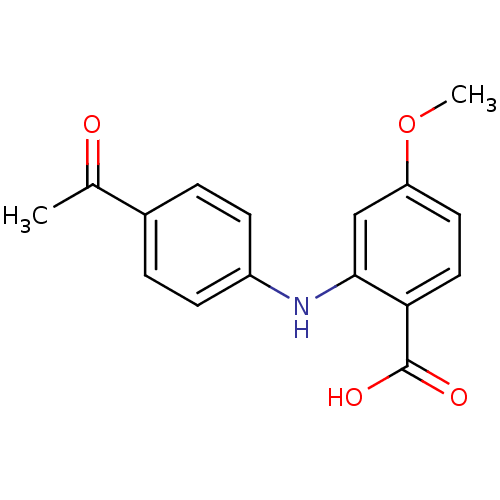
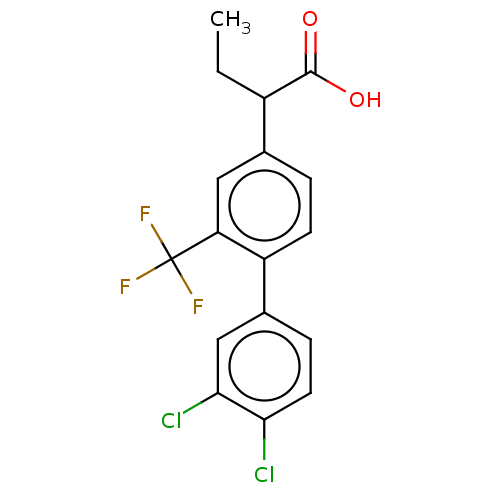
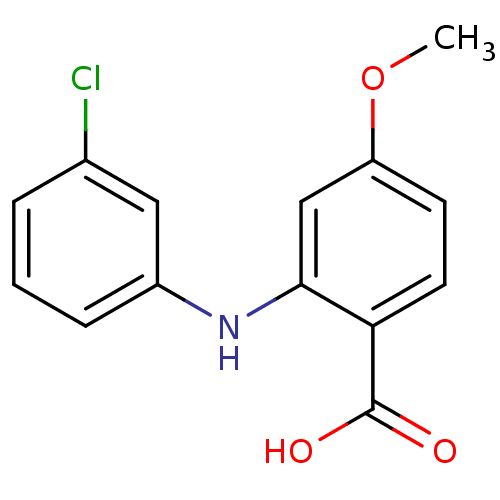
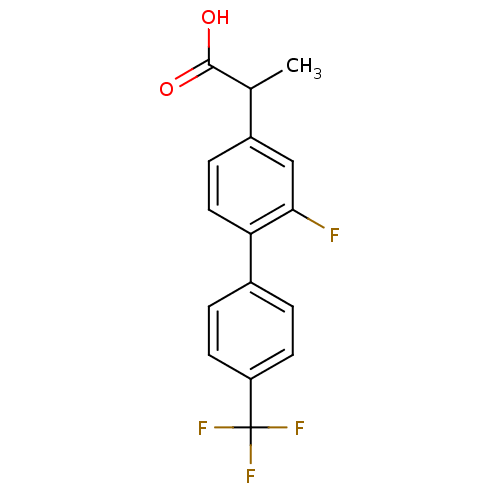
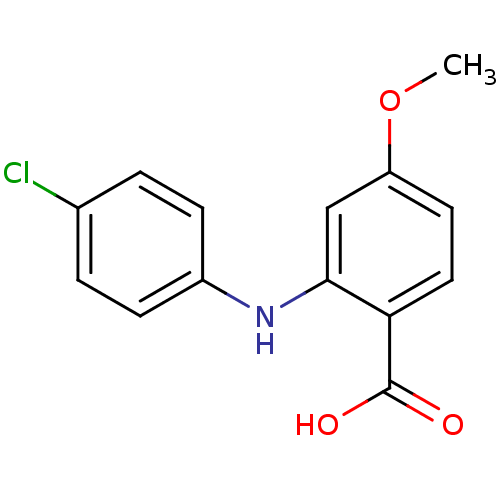
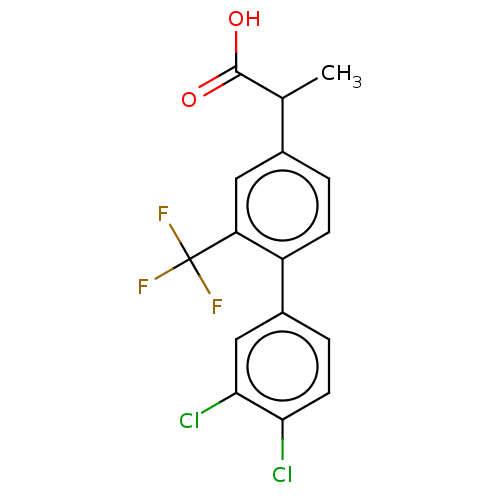
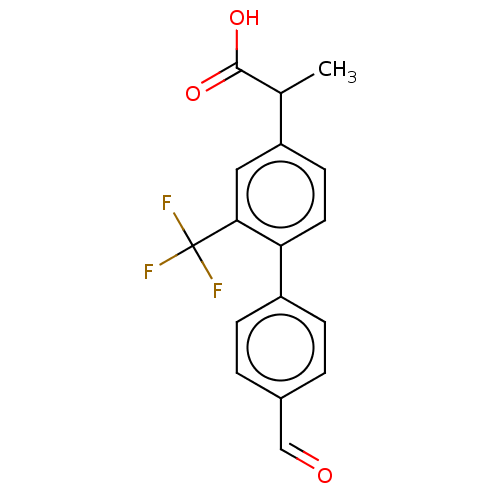
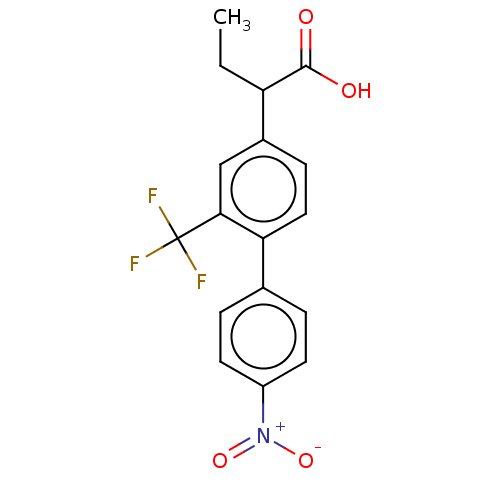
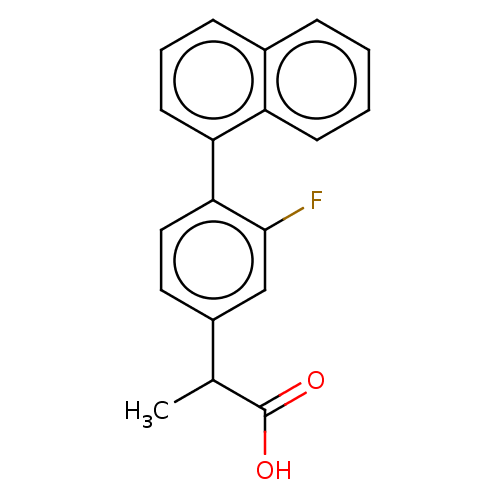
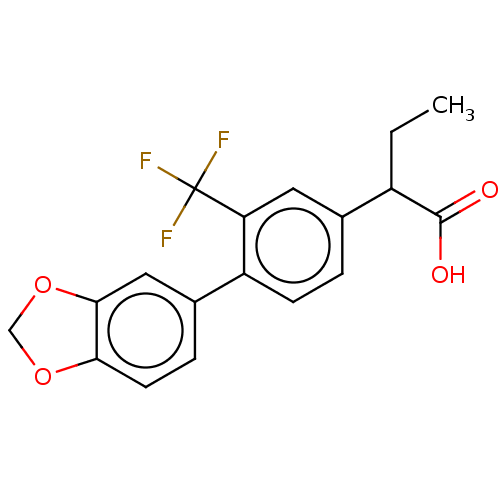
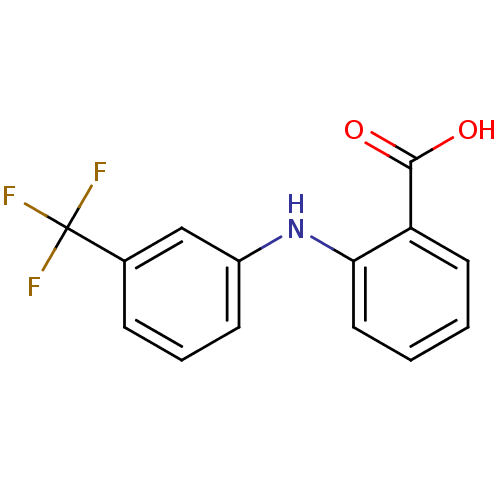
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
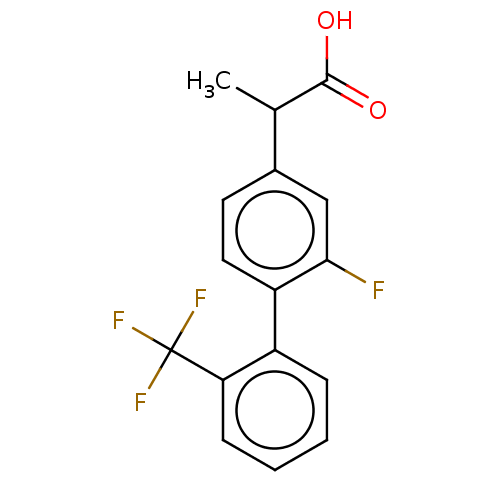
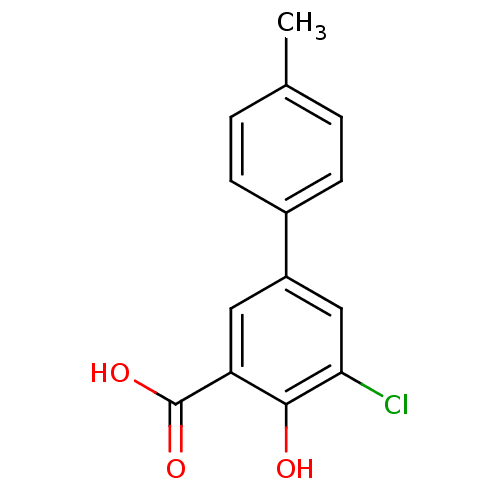
Affinity DataKi: 1.5nMAssay Description:Inhibition of human AKR1C2 dehydrogenase activity by fluorometric assayMore data for this Ligand-Target Pair
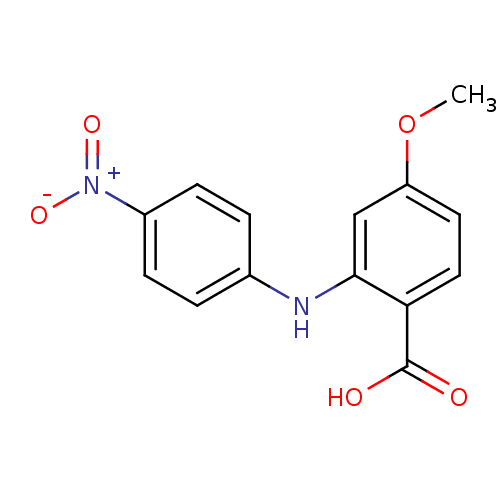
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
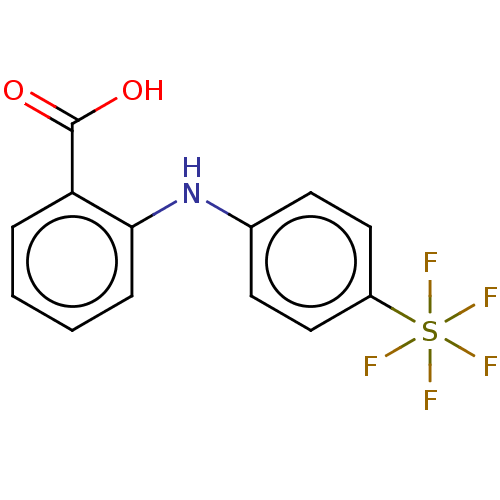
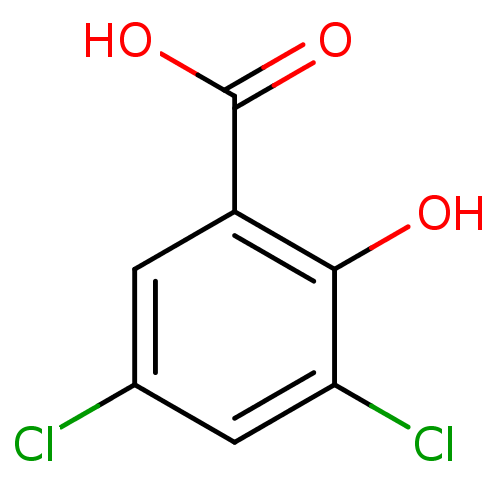
Affinity DataKi: 14nMAssay Description:Inhibition of human AKR1C2 dehydrogenase activity by fluorometric assayMore data for this Ligand-Target Pair
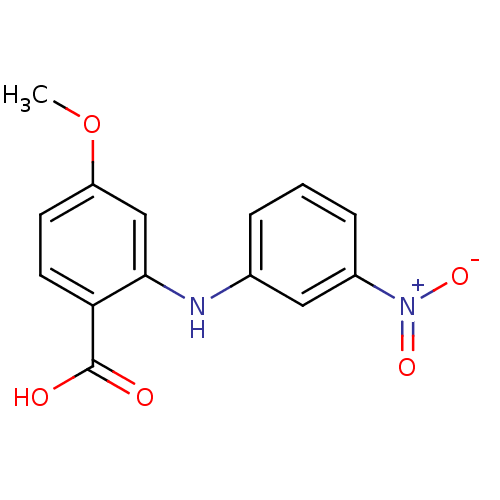
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
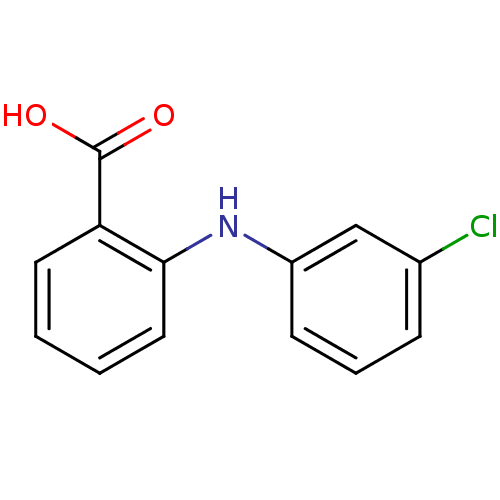
Affinity DataIC50: 14.5nMAssay Description:Inhibitors were initially screened for an ability to block the NADP+ dependent oxidation of the artificial substrate S-tetralol catalyzed by AKR1C3. ...More data for this Ligand-Target Pair
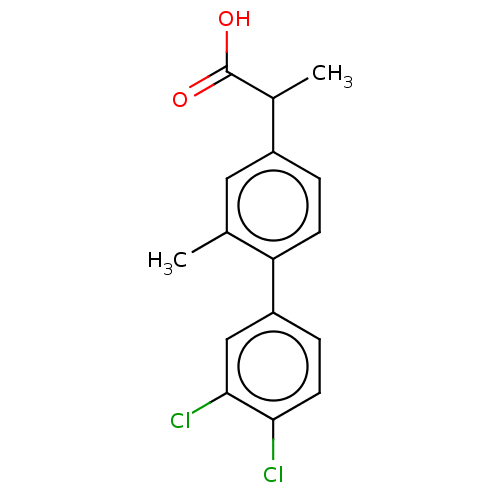
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
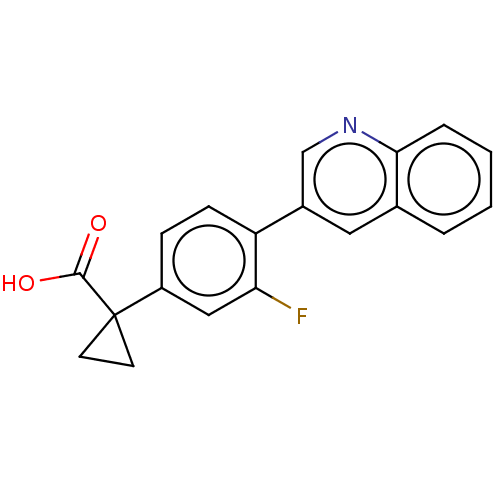
Affinity DataKi: 17nMAssay Description:Inhibition of human AKR1C2 dehydrogenase activity by fluorometric assayMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataKi: 21nMAssay Description:Inhibition of human AKR1C2 dehydrogenase activity by fluorometric assayMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataKi: 26nMAssay Description:Inhibition of human AKR1C2 dehydrogenase activity by fluorometric assayMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataKi: 29nMAssay Description:Inhibition of human AKR1C2 dehydrogenase activity by fluorometric assayMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 35.7nMAssay Description:Inhibitors were initially screened for an ability to block the NADP+ dependent oxidation of the artificial substrate S-tetralol catalyzed by AKR1C3. ...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 40.7nMAssay Description:Inhibitors were initially screened for an ability to block the NADP+ dependent oxidation of the artificial substrate S-tetralol catalyzed by AKR1C3. ...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 49.6nMAssay Description:Inhibitors were initially screened for an ability to block the NADP+ dependent oxidation of the artificial substrate S-tetralol catalyzed by AKR1C3. ...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 50.1nMAssay Description:Inhibitors were initially screened for an ability to block the NADP+ dependent oxidation of the artificial substrate S-tetralol catalyzed by AKR1C3. ...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 53.5nMAssay Description:Inhibitors were initially screened for an ability to block the NADP+ dependent oxidation of the artificial substrate S-tetralol catalyzed by AKR1C3. ...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 54.5nMAssay Description:Inhibitors were initially screened for an ability to block the NADP+ dependent oxidation of the artificial substrate S-tetralol catalyzed by AKR1C3. ...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 57nMAssay Description:Inhibitors were initially screened for an ability to block the NADP+ dependent oxidation of the artificial substrate S-tetralol catalyzed by AKR1C3. ...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataKi: 63nMAssay Description:Inhibition of human AKR1C2 dehydrogenase activity by fluorometric assayMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataKi: 70nMAssay Description:Inhibition of human AKR1C2 dehydrogenase activity by fluorometric assayMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataKi: 70nM ΔG°: -40.8kJ/molepH: 7.4 T: 2°CAssay Description:The activity was assayed by measuring the rate of change in NADPH fluorescence (at 455 nm with an excitation wavelength of 340 nm) at 298 K. When the...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 81nMAssay Description:Inhibitors were initially screened for an ability to block the NADP+ dependent oxidation of the artificial substrate S-tetralol catalyzed by AKR1C3. ...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataKi: 82nMAssay Description:Inhibition of human recombinant type 3 3-alpha-HSD expressed in Escherichia coli JM109More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataKi: 87nMAssay Description:Inhibition of human AKR1C2 dehydrogenase activity by fluorometric assayMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataKi: 87nMAssay Description:Inhibition of human recombinant type 3 3-alpha-HSD expressed in Escherichia coli JM109More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 93nMAssay Description:Inhibitors were initially screened for an ability to block the NADP+ dependent oxidation of the artificial substrate S-tetralol catalyzed by AKR1C3. ...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 100nMAssay Description:Inhibitors were initially screened for an ability to block the NADP+ dependent oxidation of the artificial substrate S-tetralol catalyzed by AKR1C3. ...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 130nMAssay Description:Inhibition of human recombinant AKR1C2 transfected in Escherichia coli BL21 (DE3) pLysS competent cells using S-tetralol as substrate assessed as inh...More data for this Ligand-Target Pair
Ligand InfoSimilars
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 150nMAssay Description:Inhibition of recombinant AKR1C2 (unknown origin) using S-tetralol as substrateMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 150nMAssay Description:Inhibition of recombinant AKR1C2 assessed as NADP+ dependent oxidation of S-tetralol by fluorescence assayMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 160nMAssay Description:Inhibition of human recombinant AKR1C2 transfected in Escherichia coli BL21 (DE3) pLysS competent cells using S-tetralol as substrate assessed as inh...More data for this Ligand-Target Pair
Ligand InfoSimilars
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataKi: 168nMAssay Description:Inhibition of human AKR1C2 dehydrogenase activity by fluorometric assayMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 190nMAssay Description:Inhibition of recombinant AKR1C2 assessed as NADP+ dependent oxidation of S-tetralol by fluorescence assayMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 190nMAssay Description:Inhibition of recombinant AKR1C2 assessed as NADP+ dependent oxidation of S-tetralol by fluorescence assayMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 200nMAssay Description:Inhibition of human recombinant AKR1C2 transfected in Escherichia coli BL21 (DE3) pLysS competent cells using S-tetralol as substrate assessed as inh...More data for this Ligand-Target Pair
Ligand InfoSimilars
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataKi: 220nMAssay Description:Inhibition of human recombinant AKR1C2 assessed as 1-acenaphthenol oxidation by spectrophotometryMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 220nMAssay Description:Inhibition of recombinant AKR1C2 assessed as NADP+ dependent oxidation of S-tetralol by fluorescence assayMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 230nMAssay Description:Inhibition of human recombinant AKR1C2 transfected in Escherichia coli BL21 (DE3) pLysS competent cells using S-tetralol as substrate assessed as inh...More data for this Ligand-Target Pair
Ligand InfoSimilars
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 240nMAssay Description:Inhibition of human recombinant AKR1C2 transfected in Escherichia coli BL21 (DE3) pLysS competent cells using S-tetralol as substrate assessed as inh...More data for this Ligand-Target Pair
Ligand InfoSimilars
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 240nMAssay Description:Inhibition of human recombinant AKR1C2 transfected in Escherichia coli BL21 (DE3) pLysS competent cells using S-tetralol as substrate assessed as inh...More data for this Ligand-Target Pair
Ligand InfoSimilars
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 240nMAssay Description:Inhibition of human recombinant AKR1C2 transfected in Escherichia coli BL21 (DE3) pLysS competent cells using S-tetralol as substrate assessed as inh...More data for this Ligand-Target Pair
Ligand InfoSimilars
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 270nMAssay Description:Inhibition of recombinant AKR1C2 (unknown origin) using S-tetralol as substrateMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 270nMAssay Description:Inhibition of recombinant AKR1C2 assessed as NADP+ dependent oxidation of S-tetralol by fluorescence assayMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 280nMAssay Description:Inhibition of recombinant AKR1C2 assessed as NADP+ dependent oxidation of S-tetralol by fluorescence assayMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 280nMAssay Description:Inhibition of human recombinant AKR1C2 transfected in Escherichia coli BL21 (DE3) pLysS competent cells using S-tetralol as substrate assessed as inh...More data for this Ligand-Target Pair
Ligand InfoSimilars
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 280nMAssay Description:Inhibition of recombinant AKR1C2 assessed as NADP+ dependent oxidation of S-tetralol by fluorescence assayMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 290nMAssay Description:Inhibition of human recombinant AKR1C2 transfected in Escherichia coli BL21 (DE3) pLysS competent cells using S-tetralol as substrate assessed as inh...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 290nMAssay Description:Inhibition of recombinant AKR1C2 assessed as NADP+ dependent oxidation of S-tetralol by fluorescence assayMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 300nMAssay Description:Inhibition of human recombinant AKR1C2 transfected in Escherichia coli BL21 (DE3) pLysS competent cells using S-tetralol as substrate assessed as inh...More data for this Ligand-Target Pair
Ligand InfoSimilars
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 310nMAssay Description:Inhibition of human recombinant AKR1C2 transfected in Escherichia coli BL21 (DE3) pLysS competent cells using S-tetralol as substrate assessed as inh...More data for this Ligand-Target Pair
Ligand InfoSimilars
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 340nMAssay Description:Inhibition of human recombinant AKR1C2 transfected in Escherichia coli BL21 (DE3) pLysS competent cells using S-tetralol as substrate assessed as inh...More data for this Ligand-Target Pair
Ligand InfoSimilars
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 350nMAssay Description:Inhibition of human recombinant AKR1C2 transfected in Escherichia coli BL21 (DE3) pLysS competent cells using S-tetralol as substrate assessed as inh...More data for this Ligand-Target Pair
Ligand InfoSimilars
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 370nMAssay Description:Inhibition of human recombinant AKR1C2 transfected in Escherichia coli BL21 (DE3) pLysS competent cells using S-tetralol as substrate assessed as inh...More data for this Ligand-Target Pair
Ligand InfoSimilars
TargetAldo-keto reductase family 1 member C2(Human)
Monash University (Parkville Campus)
Curated by ChEMBL
Monash University (Parkville Campus)
Curated by ChEMBL
Affinity DataIC50: 370nMAssay Description:Inhibition of AKR1C2 by fluorimetric methodMore data for this Ligand-Target Pair
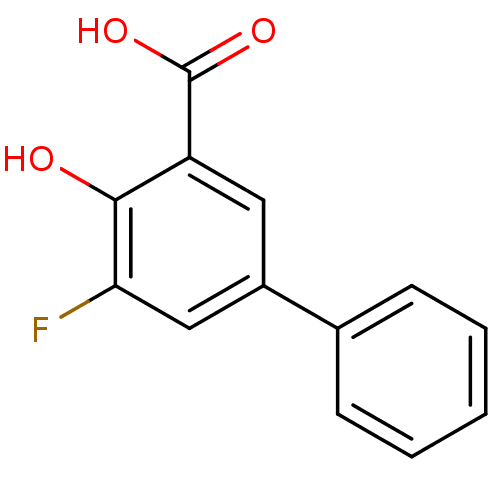
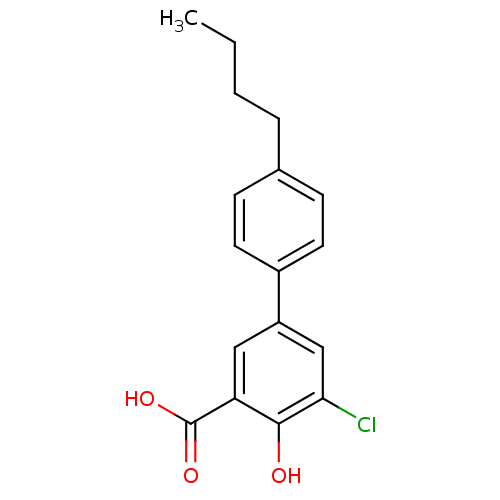
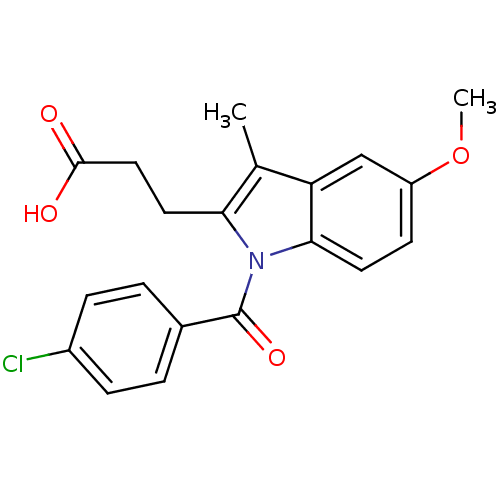
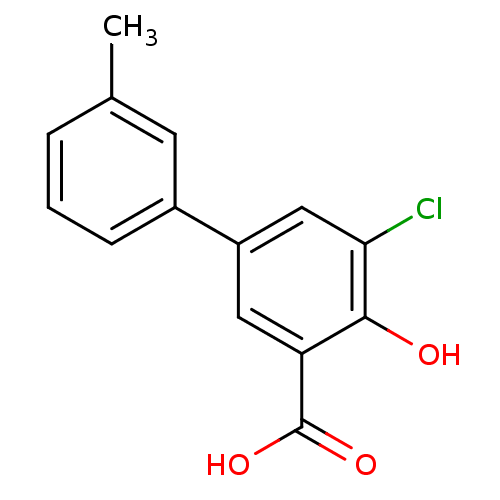
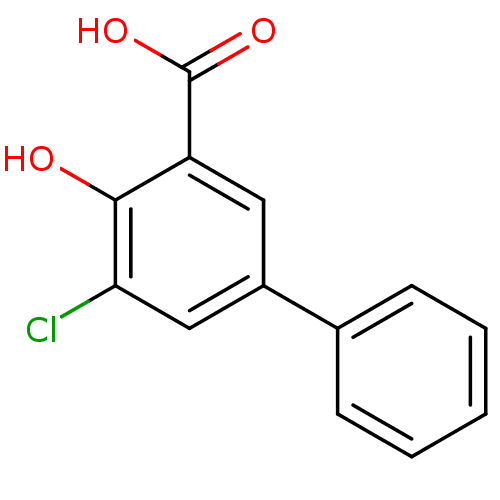
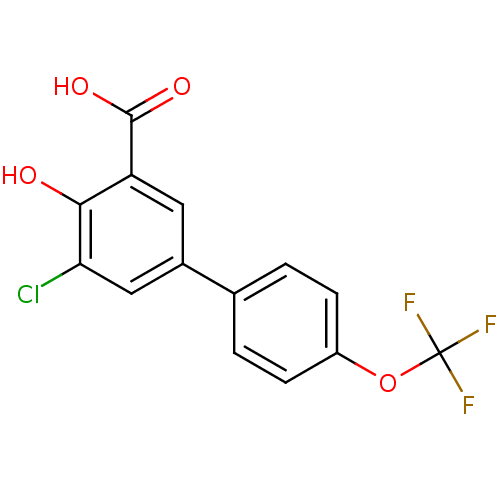
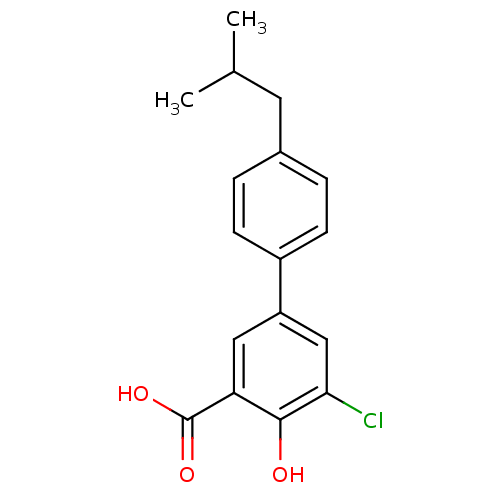
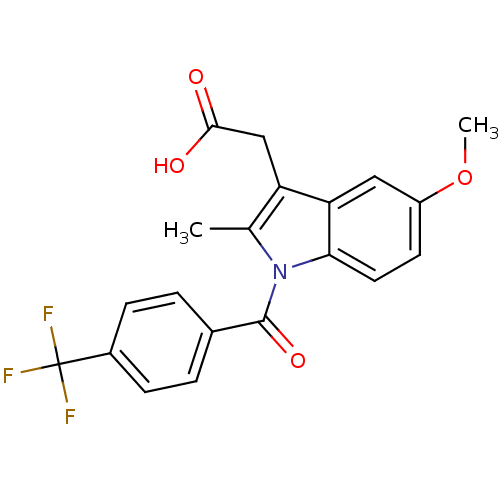
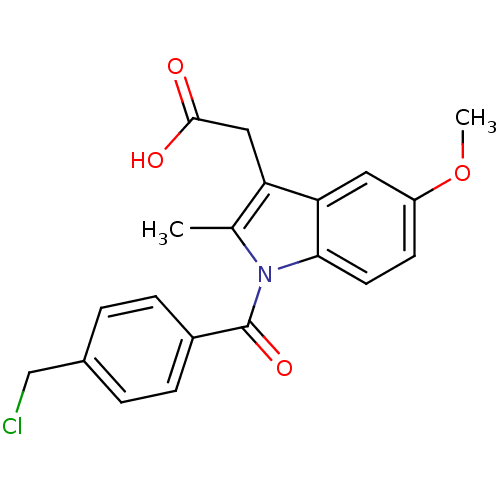
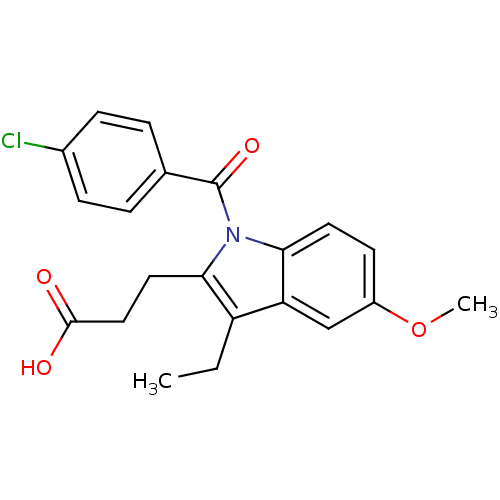
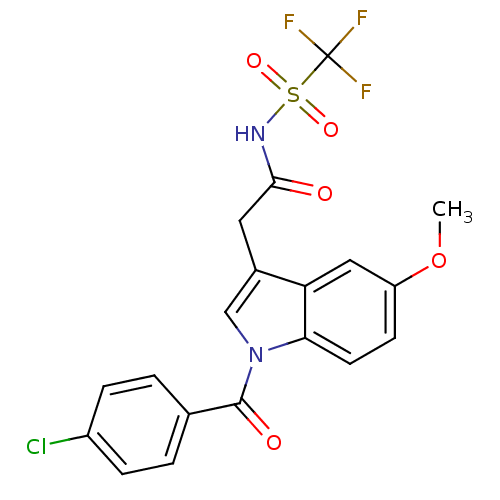
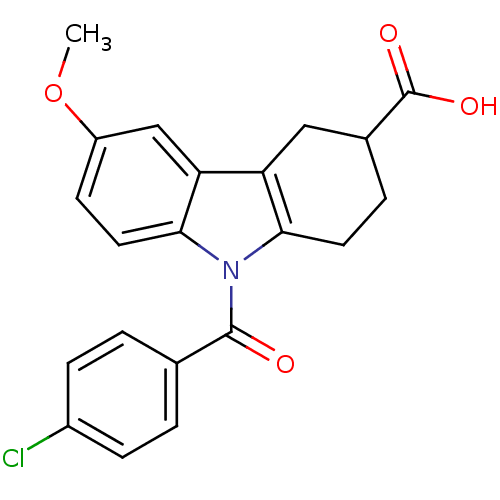
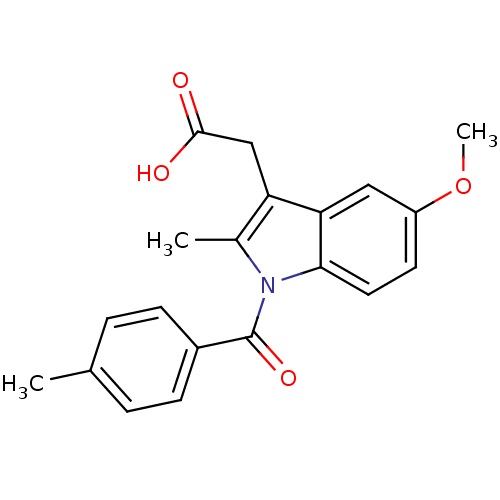
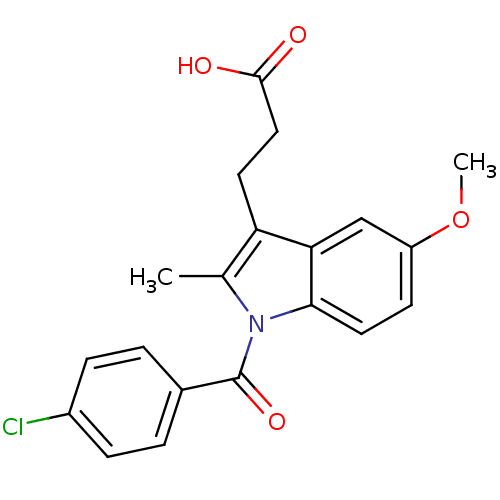
 3D Structure (crystal)
3D Structure (crystal)