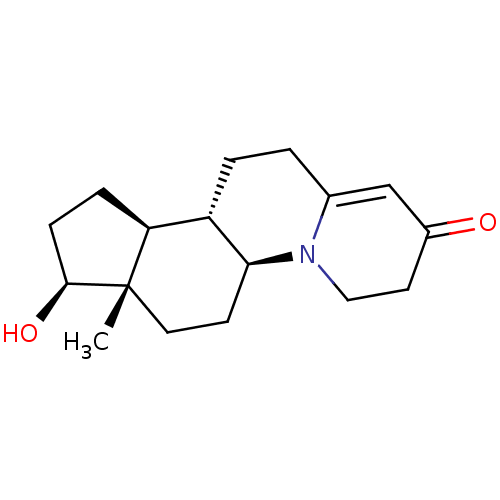
BDBM50057297 (5aS,7aS,8S,10aS,10bS)-8-Hydroxy-7a-methyl-3,4,6,7,7a,8,9,10,10a,10b,11,12-dodecahydro-5aH-indeno[4,5-c]quinolizin-2-one::CHEMBL269632
SMILES C[C@]12CC[C@H]3[C@@H](CCC4=CC(=O)CCN34)[C@@H]1CC[C@@H]2O
InChI Key InChIKey=UTDXFFWXJOMGPG-UHFFFAOYSA-N
Data 3 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50057297
Found 3 hits for monomerid = 50057297
Affinity DataIC50: 2.70nMAssay Description:Inhibition against 5 alpha R-2 in human prostate homogenate relative to finasterideMore data for this Ligand-Target Pair
Affinity DataIC50: 409nMAssay Description:Inhibitory activity against human 5-alpha Reductase-1 expressed in DU-145 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 4.20E+3nMAssay Description:Inhibitory activity against 5-alpha Reductase-2 on human prostate homogenates from surgically derived benign hyperplastic tissueMore data for this Ligand-Target Pair