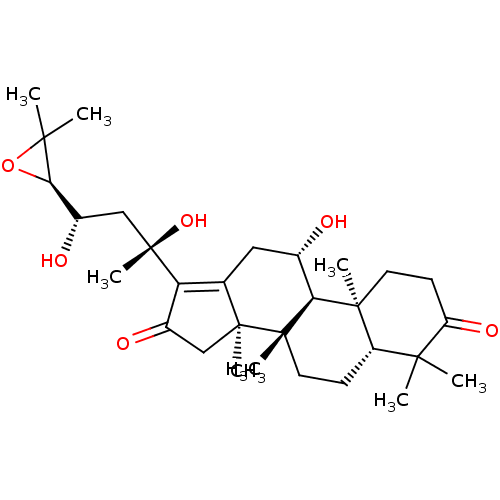
BDBM50130916 CHEMBL3632945
SMILES [H][C@@]1(OC1(C)C)[C@@H](O)C[C@](C)(O)C1=C2C[C@H](O)[C@@]3([H])[C@@]4(C)CCC(=O)C(C)(C)[C@]4([H])CC[C@]3(C)[C@@]2(C)CC1=O
InChI Key InChIKey=SSNKUHZPKQIDOG-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 2 hits for monomerid = 50130916
Found 2 hits for monomerid = 50130916
Affinity DataEC50: 4.07E+4nMAssay Description:Transactivation of FXR (unknown origin) transfected in HepG2 cells co-expressing pBSEP/pGL4.74 incubated for 24 hrs by luciferase reporter gene assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+5nMAssay Description:Inhibition of human carboxylesterase 2 using 4-benzoyl-N-butyl-1,8-naphthalimide as substrate by fluorescence assayMore data for this Ligand-Target Pair