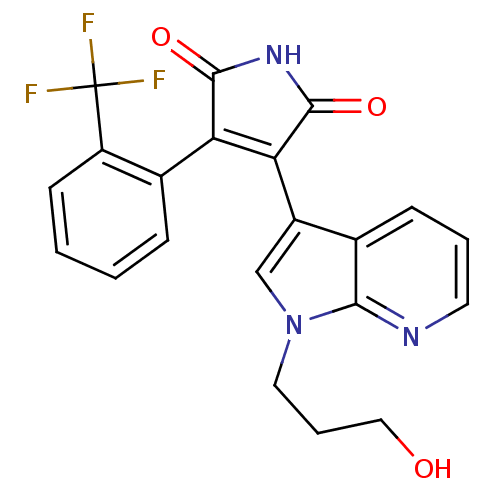
BDBM50147461 3-[1-(3-Hydroxy-propyl)-1H-pyrrolo[2,3-b]pyridin-3-yl]-4-(2-trifluoromethyl-phenyl)-pyrrole-2,5-dione::CHEMBL325817
SMILES OCCCn1cc(C2=C(C(=O)NC2=O)c2ccccc2C(F)(F)F)c2cccnc12
InChI Key InChIKey=PLUWMEIYYZQPRH-UHFFFAOYSA-N
Data 3 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50147461
Found 3 hits for monomerid = 50147461
TargetGlycogen synthase kinase-3 beta(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 14nMAssay Description:Inhibitory concentration against rabbit glycogen synthase kinase-3 beta using protein phosphatase inhibitor-2 as substrateMore data for this Ligand-Target Pair
TargetGlycogen synthase kinase-3 beta(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 14nMAssay Description:Inhibition of full length recombinant His-tagged human Glycogen synthase kinase-3 beta expressed in Baculovirus expression systemMore data for this Ligand-Target Pair
TargetProtein kinase C beta type(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 1.85E+3nMAssay Description:Inhibitory concentration against human protein kinase C-betaII using histone as substrateMore data for this Ligand-Target Pair