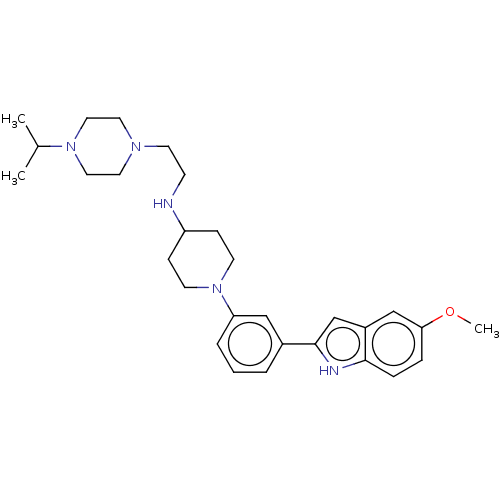
BDBM50158907 CHEMBL3786444::US11247985, Table 3.10
SMILES COc1ccc2[nH]c(cc2c1)-c1cccc(c1)N1CCC(CC1)NCCN1CCN(CC1)C(C)C
InChI Key InChIKey=LIEFKOLTZCXUOY-UHFFFAOYSA-N
Data 2 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 2 hits for monomerid = 50158907
Found 2 hits for monomerid = 50158907
Affinity DataIC50: 660nMAssay Description:To optimize p97 inhibitors, the C-5 trifluoromethylated trifluoromethylated indole 12 was generated as a promising lead structure. In the ADP-Glo ass...More data for this Ligand-Target Pair
Affinity DataIC50: 710nMAssay Description:Inhibition of wild type human AAA ATPase p97 expressed in Escherichia coli measured by ADPGlo assay in presence of 100 uM ATPMore data for this Ligand-Target Pair