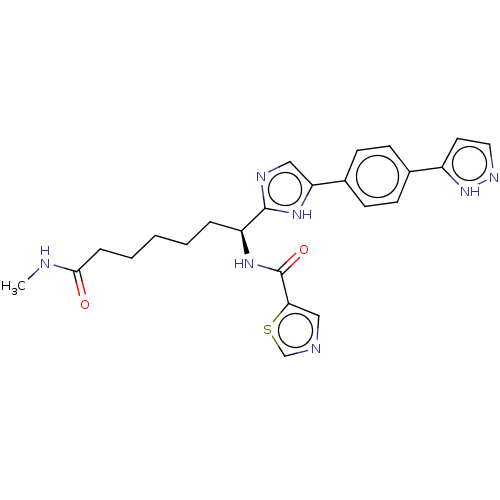
BDBM50175035 CHEMBL3808866
SMILES CNC(=O)CCCCC[C@H](NC(=O)c1cncs1)c1ncc([nH]1)-c1ccc(cc1)-c1ccn[nH]1
InChI Key InChIKey=UFQDUCIOZJEAJB-UHFFFAOYSA-N
Data 3 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50175035
Found 3 hits for monomerid = 50175035
Affinity DataIC50: 330nMAssay Description:Inhibition of human HDAC3More data for this Ligand-Target Pair
Affinity DataIC50: 1.85E+3nMAssay Description:Inhibition of recombinant human His-tagged HDAC1 expressed in insect cells preincubated for 10 mins followed by addition of FLUOR DE LYS as fluoresce...More data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+4nMAssay Description:Inhibition of human ERG after 4 hrs by fluorescence polarization assayMore data for this Ligand-Target Pair