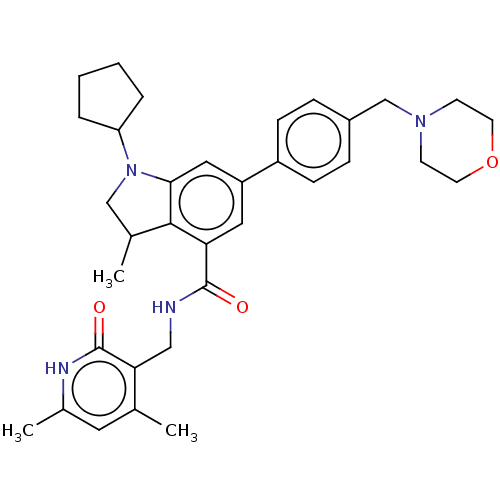
BDBM50205636 CHEMBL3923183
SMILES CC1CN(C2CCCC2)c2cc(cc(C(=O)NCc3c(C)cc(C)[nH]c3=O)c12)-c1ccc(CN2CCOCC2)cc1
InChI Key InChIKey=QHNITKGBPYIHKK-UHFFFAOYSA-N
Data 3 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50205636
Found 3 hits for monomerid = 50205636
Affinity DataIC50: 448nMAssay Description:Inhibition of human wild type EZH2 using histone H3 as substrate after 1 hr in presence of 3H-SAM by filter paper detection analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 448nMAssay Description:Inhibition of human wild type EZH2 using histone H3 as substrate after 1 hr in presence of 3H-SAM by filter paper detection analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 2.65E+3nMAssay Description:Inhibition of human EZH1 complex using histone H3 as substrate after 1 hr in presence of 3H-SAM by filter paper detection analysisMore data for this Ligand-Target Pair