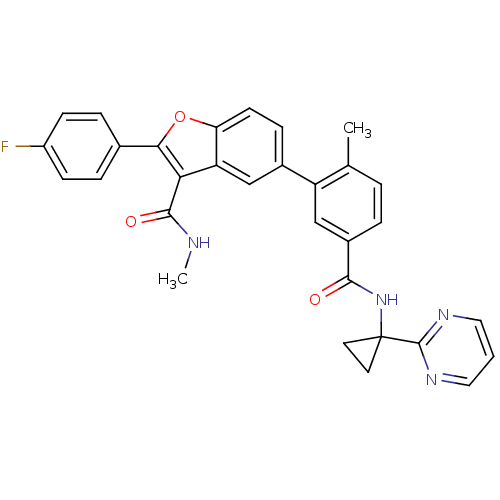
BDBM50239249 CHEMBL4104000
SMILES CNC(=O)c1c(oc2ccc(cc12)-c1cc(ccc1C)C(=O)NC1(CC1)c1ncccn1)-c1ccc(F)cc1
InChI Key InChIKey=LUIDEMBRIXHMJX-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 50239249
Found 4 hits for monomerid = 50239249
Affinity DataIC50: 3.90E+3nMAssay Description:Inhibition of microsomal CYP2C8 (unknown origin)More data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Human)
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Affinity DataIC50: 4.80E+3nMAssay Description:Inhibition of human ERG by thallium flux assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.14E+4nMAssay Description:Inhibition of microsomal CYP3A4 (unknown origin)More data for this Ligand-Target Pair
TargetNuclear receptor subfamily 1 group I member 2(Human)
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
Affinity DataEC50: >5.00E+4nMAssay Description:Transactivation of PXR (unknown origin)More data for this Ligand-Target Pair