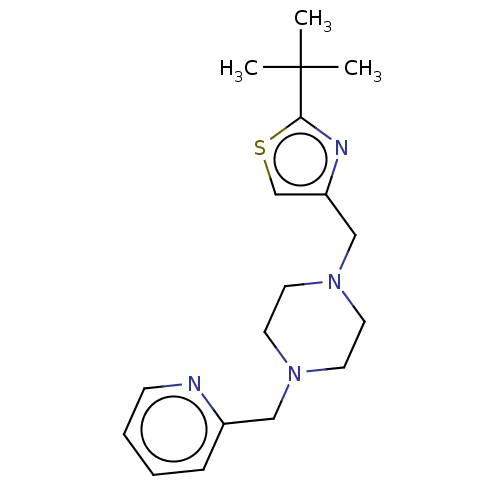
BDBM50281392 CHEMBL4164366
SMILES CC(C)(C)c1nc(CN2CCN(Cc3ccccn3)CC2)cs1
InChI Key InChIKey=IWGYSGOGENBEPJ-UHFFFAOYSA-N
Data 3 KI
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50281392
Found 3 hits for monomerid = 50281392
Affinity DataKi: 1.19E+3nMAssay Description:Displacement of [3H]pyrilamine from human HRH1 expressed in HEK cell membranes after 90 mins by scintillation counting methodMore data for this Ligand-Target Pair
Affinity DataKi: >1.00E+4nMAssay Description:Displacement of [3H]ketanserin from 5-HT2A (unknown origin) expressed in membranes after 90 mins by scintillation counting methodMore data for this Ligand-Target Pair
Affinity DataKi: >1.00E+4nMAssay Description:Displacement of [3H]N-methylspiperone from human DRD2 after 90 mins by scintillation counting methodMore data for this Ligand-Target Pair