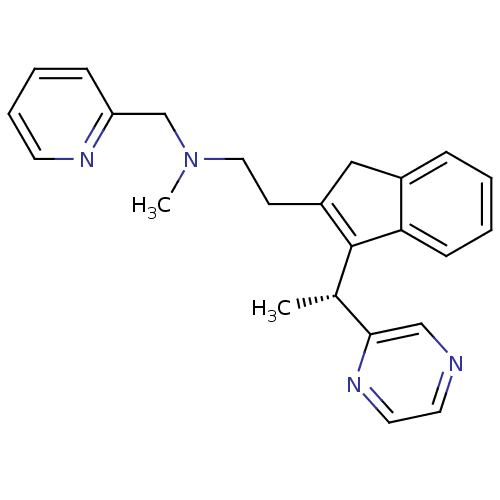
BDBM50336085 (R)-N-methyl-2-(3-(1-(pyrazin-2-yl)ethyl)-1H-inden-2-yl)-N-(pyridin-2-ylmethyl)ethanamine::CHEMBL1669424
SMILES C[C@H](C1=C(CCN(C)Cc2ccccn2)Cc2ccccc12)c1cnccn1
InChI Key InChIKey=IJFPVAYNHAMKAK-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50336085
Found 3 hits for monomerid = 50336085
Affinity DataKi: 13.9nMAssay Description:Binding affinity to histamine H1 receptorMore data for this Ligand-Target Pair
Affinity DataIC50: 4.78E+3nMAssay Description:Inhibition of CYP3A4More data for this Ligand-Target Pair
Affinity DataIC50: 5.71E+3nMAssay Description:Inhibition of CYP2D6More data for this Ligand-Target Pair