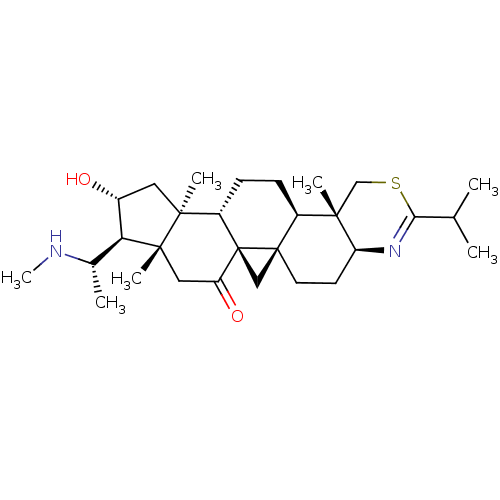
BDBM50346349 16alpha-Hydroxy-4beta,14alpha-dimethyl-20S-(methylamino)-9,19-cyclo-{2'-isopropyl-5',6'-dihydro-4'alphaH-[1',3']thiazino[4',5':3,4]}-5alpha,9beta-pregnan-11-one::CHEMBL1783521
SMILES CN[C@@H](C)[C@H]1[C@H](O)C[C@@]2(C)[C@@H]3CC[C@@H]4[C@]5(C[C@@]35C(=O)C[C@]12C)CC[C@@H]1N=C(SC[C@@]41C)C(C)C
InChI Key InChIKey=XMAZYYDJYBXRBC-UHFFFAOYSA-N
Data 3 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50346349
Found 3 hits for monomerid = 50346349
Affinity DataIC50: 45nMAssay Description:Inhibition of Electrophorus electricus AChE using acetylthiocholine iodide as substrate by spectroscopic Ellmans methodMore data for this Ligand-Target Pair
Affinity DataIC50: 76nMAssay Description:Inhibition of recombinant human AChE using acetylthiocholine iodide as substrate by spectroscopic Ellmans methodMore data for this Ligand-Target Pair
Affinity DataIC50: 1.34E+3nMAssay Description:Inhibition of recombinant human serum BChE using butyrylthiocholine iodide as substrate by spectroscopic Ellmans methodMore data for this Ligand-Target Pair