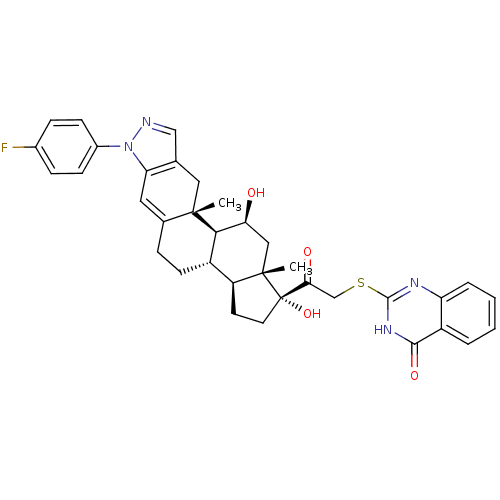
BDBM50381745 CHEMBL2023621
SMILES C[C@]12C[C@H](O)[C@H]3[C@@H](CCC4=Cc5c(C[C@]34C)cnn5-c3ccc(F)cc3)[C@@H]1CC[C@]2(O)C(=O)CSc1nc2ccccc2c(=O)[nH]1
InChI Key InChIKey=UVYKDWJUINPICB-UHFFFAOYSA-N
Data 3 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50381745
Found 3 hits for monomerid = 50381745
Affinity DataIC50: 3nMAssay Description:Transrepression activity at glucocorticoid receptor in human H292 cells assessed as inhibition of TNF-induced IL8 productionMore data for this Ligand-Target Pair
Affinity DataIC50: 86.5nMAssay Description:Transactivation activity at glucocorticoid receptor in rat H4II-E cells assessed as induction of tyrosine aminotransferaseMore data for this Ligand-Target Pair
Affinity DataIC50: 93.5nMAssay Description:Transactivation activity at glucocorticoid receptor in human HepG2 cells assessed as induction of tyrosine aminotransferaseMore data for this Ligand-Target Pair