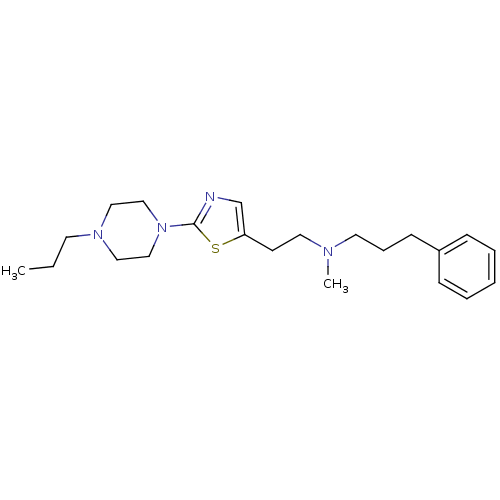
BDBM50413824 CHEMBL473093
SMILES CCCN1CCN(CC1)c1ncc(CCN(C)CCCc2ccccc2)s1
InChI Key InChIKey=LHFFBQAOSCEDBC-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50413824
Found 3 hits for monomerid = 50413824
Affinity DataKi: 3.20nMAssay Description:Displacement of [3H]-Nalpha-methylhistamine from human histamine H3 receptor expressed in HEK293T cell membranes after 60 mins by liquid scintillatio...More data for this Ligand-Target Pair
Affinity DataIC50: 1.40E+4nMAssay Description:Inhibition of Electrophorus electricus AchE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition and me...More data for this Ligand-Target Pair
Affinity DataIC50: 1.46E+4nMAssay Description:Inhibition of equine serum BuChE using butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition and measured aft...More data for this Ligand-Target Pair