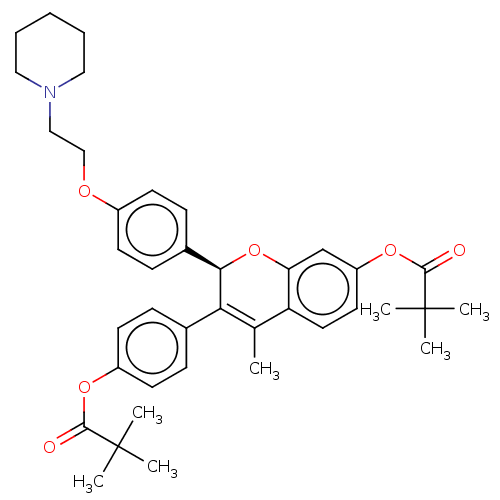
BDBM50471253 CHEMBL441622
SMILES CC1=C([C@H](Oc2cc(OC(=O)C(C)(C)C)ccc12)c1ccc(OCCN2CCCCC2)cc1)c1ccc(OC(=O)C(C)(C)C)cc1
InChI Key InChIKey=OEKMGABCSLYWOP-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50471253
Found 3 hits for monomerid = 50471253
Affinity DataKi: 4.20nMAssay Description:Apparent binding affinity against estradiol-stimulated T-47D cell proliferationMore data for this Ligand-Target Pair
Affinity DataIC50: 36nMAssay Description:Inhibition of estradiol-stimulated T-47D cell proliferation.More data for this Ligand-Target Pair
Affinity DataKi: >270nMAssay Description:Inhibition of estradiol binding to estrogen receptor in Human Breast cancer cytosol (3.3% ethanol)More data for this Ligand-Target Pair