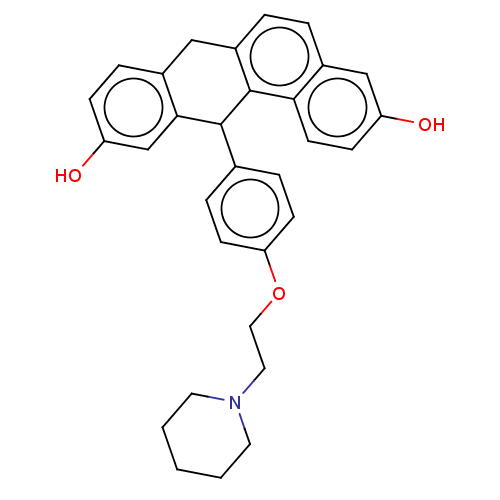
BDBM50474891 CHEMBL184423
SMILES Oc1ccc2Cc3ccc4cc(O)ccc4c3C(c3ccc(OCCN4CCCCC4)cc3)c2c1
InChI Key InChIKey=OEVSLFVSLIUCMB-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50474891
Found 3 hits for monomerid = 50474891
Affinity DataEC50: 2.60nMAssay Description:Agonist activity as alkaline phosphatase induction in Ishikawa endometrial cells compared to E2More data for this Ligand-Target Pair
Affinity DataIC50: 6.60nMAssay Description:Antagonist effect against 10 pM 17-beta-estradiol induced MCF-7 cell proliferationMore data for this Ligand-Target Pair
Affinity DataIC50: 130nMAssay Description:Antagonist activity as inhibition of 1 nM 17-beta-estradiol stimulated alkaline phosphatase induction in Ishikawa endometrial cellsMore data for this Ligand-Target Pair