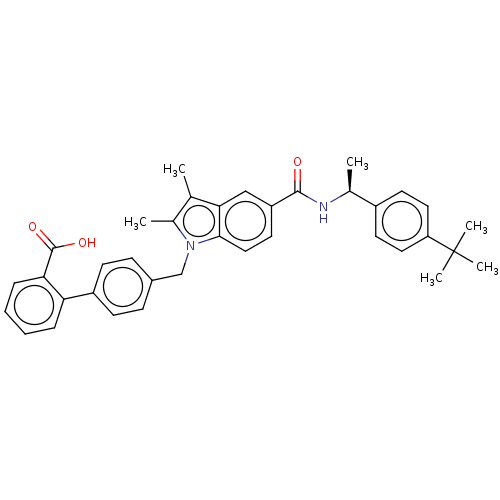
BDBM50498890 CHEMBL3735924::US10744117, Compound IB-2
SMILES C[C@H](NC(=O)c1ccc2n(Cc3ccc(cc3)-c3ccccc3C(O)=O)c(C)c(C)c2c1)c1ccc(cc1)C(C)(C)C
InChI Key InChIKey=WKDKAVKAQXVFJV-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50498890
Found 3 hits for monomerid = 50498890
TargetPeroxisome proliferator-activated receptor gamma(Human)
The Scripps Research Institute
Curated by ChEMBL
The Scripps Research Institute
Curated by ChEMBL
Affinity DataIC50: 5nMAssay Description:Binding affinity to GST-tagged PPAR-gamma-LBD (unknown origin) after 2 hrs by Lantha screen assay using fluormone Pan-PPAR green probeMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
The Scripps Research Institute
Curated by ChEMBL
The Scripps Research Institute
Curated by ChEMBL
Affinity DataEC50: 355nMAssay Description:Transactivation of human PPARgamma in HEK293T cells after 18 hrs by luciferase reporter gene assayMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
The Scripps Research Institute
Curated by ChEMBL
The Scripps Research Institute
Curated by ChEMBL