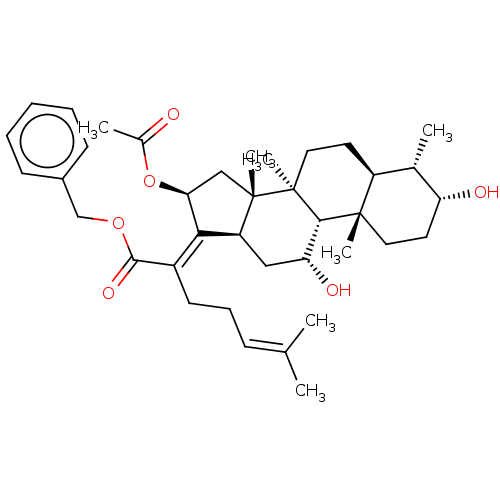
BDBM50515967 CHEMBL4439071
SMILES [H][C@@]12[#6]-[#6@@H](-[#8])[C@@]3([H])[C@@]4([#6])[#6]-[#6]-[#6@@H](-[#8])-[#6@@H](-[#6])[C@]4([H])[#6]-[#6][C@]3([#6])[C@@]1([#6])[#6]-[#6@H](-[#8]-[#6](-[#6])=O)\[#6]2=[#6](\[#6]-[#6]\[#6]=[#6](/[#6])-[#6])-[#6](=O)-[#8]-[#6]-c1ccccc1
InChI Key InChIKey=QFALWWDHMQCEGC-UHFFFAOYSA-N
Data 6 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 6 hits for monomerid = 50515967
Found 6 hits for monomerid = 50515967
Affinity DataIC50: 7nMAssay Description:Reversal of P-gp-mediated multidrug resistance in human KBV cells assessed as potentiation of paclitaxel-induced cytotoxicity by measuring paclitaxel...More data for this Ligand-Target Pair
Affinity DataIC50: 17nMAssay Description:Reversal of P-gp-mediated multidrug resistance in human KBV cells assessed as potentiation of paclitaxel-induced cytotoxicity by measuring paclitaxel...More data for this Ligand-Target Pair
Affinity DataIC50: 92nMAssay Description:Reversal of P-gp-mediated multidrug resistance in human KBV cells assessed as potentiation of vincristine-induced cytotoxicity by measuring vincristi...More data for this Ligand-Target Pair
Affinity DataIC50: 144nMAssay Description:Reversal of P-gp-mediated multidrug resistance in human KBV cells assessed as potentiation of vincristine-induced cytotoxicity by measuring vincristi...More data for this Ligand-Target Pair
Affinity DataIC50: 1.62E+4nMAssay Description:Reversal of P-gp-mediated multidrug resistance in human KBV cells assessed as potentiation of irinotecan-induced cytotoxicity by measuring irinotecan...More data for this Ligand-Target Pair
Affinity DataIC50: 3.73E+4nMAssay Description:Reversal of P-gp-mediated multidrug resistance in human KBV cells assessed as potentiation of irinotecan-induced cytotoxicity by measuring irinotecan...More data for this Ligand-Target Pair