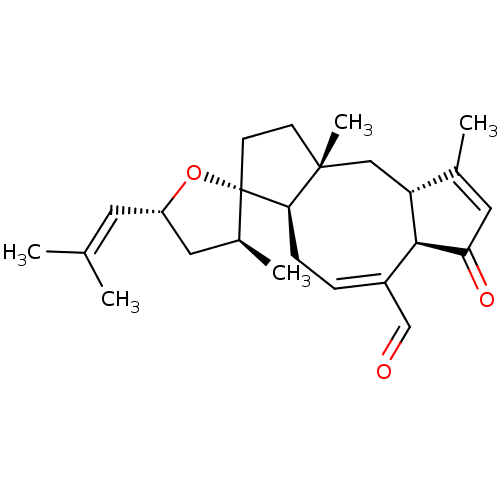
BDBM50530670 3-ANHYDRO-6-EPI-OPHIOBOLIN A
SMILES [H][C@]12[#6][C@@]3([#6])[#6]-[#6][C@@]4([#8]-[#6@H](-[#6]-[#6@@H]4-[#6])\[#6]=[#6](\[#6])-[#6])[C@]3([H])[#6]\[#6]=[#6](-[#6]=O)/[C@]1([H])[#6](=O)-[#6]=[#6]2-[#6]
InChI Key InChIKey=HJVZKYVURJEXEK-UHFFFAOYSA-N
Data 2 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 2 hits for monomerid = 50530670
Found 2 hits for monomerid = 50530670
Target3-hydroxy-3-methylglutaryl-coenzyme A reductase(Human)
Huazhong University of Science and Technology
Curated by ChEMBL
Huazhong University of Science and Technology
Curated by ChEMBL
Affinity DataIC50: 8.40E+3nMAssay Description:Inhibition of human HMGR using HMGCoA as substrate measured after 15 mins in presence of NADPH by UV microplate reader analysisMore data for this Ligand-Target Pair
Target3-hydroxy-3-methylglutaryl-coenzyme A reductase(Human)
Huazhong University of Science and Technology
Curated by ChEMBL
Huazhong University of Science and Technology
Curated by ChEMBL
Affinity DataIC50: 8.40E+3nMAssay Description:Inhibition of human HMGR using HMGCoA as substrate measured after 15 mins in presence of NADPH by UV microplate reader analysisMore data for this Ligand-Target Pair