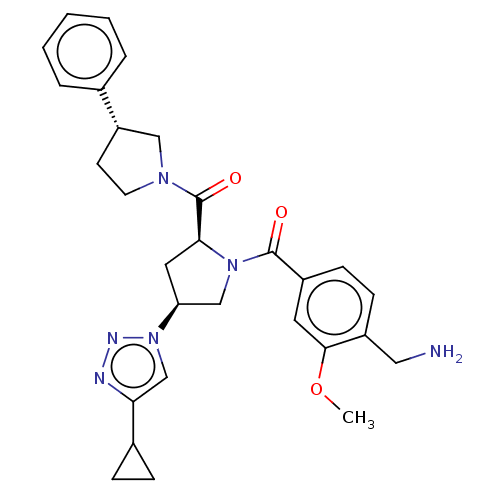
BDBM50533266 CHEMBL4475317
SMILES COc1cc(ccc1CN)C(=O)N1C[C@H](C[C@H]1C(=O)N1CC[C@@H](C1)c1ccccc1)n1cc(nn1)C1CC1
InChI Key InChIKey=UOZIVEGXVLHOBN-UHFFFAOYSA-N
Data 4 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 50533266
Found 4 hits for monomerid = 50533266
Affinity DataIC50: 220nMAssay Description:Inhibition of human PRSS1 assessed as enzymatic cleavage of Benzoyl-GlyPro-Arg'Rh110-gammaGlu-OH by fluorescence analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 220nMAssay Description:Inhibition of human PRSS1 assessed as enzymatic cleavage of Benzoyl-GlyPro-Arg'Rh110-gammaGlu-OH by fluorescence analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 1.40E+4nMAssay Description:Displacement of [3H]-dofetilide from human ERG expressed in HEK293 cells measured after 90 mins by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.40E+4nMAssay Description:Displacement of [3H]-dofetilide from human ERG expressed in HEK293 cells measured after 90 mins by scintillation proximity assayMore data for this Ligand-Target Pair