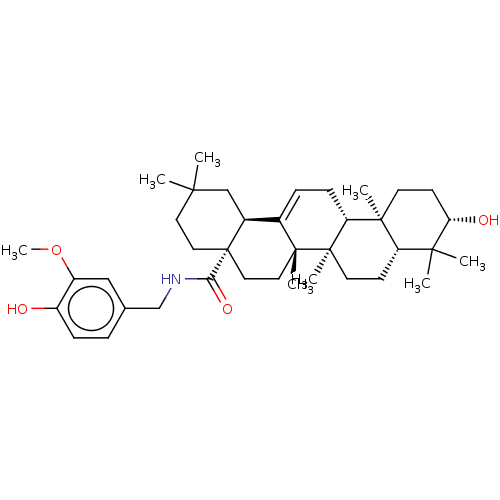
BDBM50554642 CHEMBL4800169
SMILES [H][C@@]12CC(C)(C)CC[C@@]1(CC[C@]1(C)C2=CC[C@]2([H])[C@@]3(C)CC[C@H](O)C(C)(C)[C@]3([H])CC[C@@]12C)C(=O)NCc1ccc(O)c(OC)c1
InChI Key InChIKey=STERUNUCURXMBK-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 2 hits for monomerid = 50554642
Found 2 hits for monomerid = 50554642
TargetTransient receptor potential cation channel subfamily V member 1(Human)
National Research Council (Icb-Cnr)
Curated by ChEMBL
National Research Council (Icb-Cnr)
Curated by ChEMBL
Affinity DataEC50: 35nMAssay Description:Agonist activity at human TRPV1 stably transfected in HEK293 cells assessed as increase in calcium influx in presence of ionomycin by Fluo-4-AM dye b...More data for this Ligand-Target Pair
TargetTransient receptor potential cation channel subfamily V member 1(Human)
National Research Council (Icb-Cnr)
Curated by ChEMBL
National Research Council (Icb-Cnr)
Curated by ChEMBL
Affinity DataIC50: 50nMAssay Description:Antagonist activity at human TRPV1 stably transfected in HEK293 cells assessed as decrease in calcium influx preincubated for 5 mins followed by caps...More data for this Ligand-Target Pair