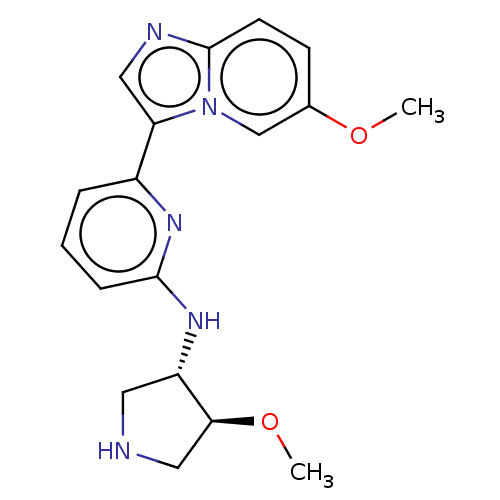
BDBM621095 6-(6-methoxyimidazo- [1,2-a]pyridin-3-yl)- N-((3S,4S)-4-methoxy- pyrrolidin-3-yl)pyridin- 2-amine::US20230303563, Compound 45
SMILES CO[C@H]1CNC[C@@H]1Nc1cccc(n1)-c1cnc2ccc(OC)cn12
InChI Key InChIKey=PQMBTEFIJYDBHA-UHFFFAOYSA-N
Data 3 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 621095
Found 3 hits for monomerid = 621095
Affinity DataIC50: 0.5nMAssay Description:Table 32: The reagent used was as follows: Base Reaction buffer; 20 mM Hepes (pH 7.5), 10 mM MgCl2, 1 mM EGTA, 0.016 Brij35, 0.02 mg/ml BSA, 0.1 mM N...More data for this Ligand-Target Pair
Affinity DataIC50: 3nMAssay Description:Table 32: The reagent used was as follows: Base Reaction buffer; 20 mM Hepes (pH 7.5), 10 mM MgCl2, 1 mM EGTA, 0.016 Brij35, 0.02 mg/ml BSA, 0.1 mM N...More data for this Ligand-Target Pair
Affinity DataIC50: 492nMAssay Description:Table 32: The reagent used was as follows: Base Reaction buffer; 20 mM Hepes (pH 7.5), 10 mM MgCl2, 1 mM EGTA, 0.016 Brij35, 0.02 mg/ml BSA, 0.1 mM N...More data for this Ligand-Target Pair